An atom is the smallest piece of matter that has the characteristic properties of a chemical element, such as hydrogen, oxygen, calcium, iron, gold, and neon. More than 90 types of atom exist in nature, and each one forms a different element. Elements are made up of only one type of atom—gold contains only gold atoms, and neon contains only.

The atom is the basic building block for all matter in the universe. Atoms are extremely small and are made up of a few even smaller particles. The basic particles that make up an atom are electrons, protons, and neutrons. Atoms fit together with other atoms to make up matter. It takes a lot of atoms to make up anything. There are so many atoms.

Build an Atom - Inquiry-based basics (homework version) Description Updated 2020 for the HTML sim. This could be used for a homework for a traditional class or an online course. There are slides that could be used for poling students after the activity. Some of the materials were designed for the Java sim, so some images may be different from.

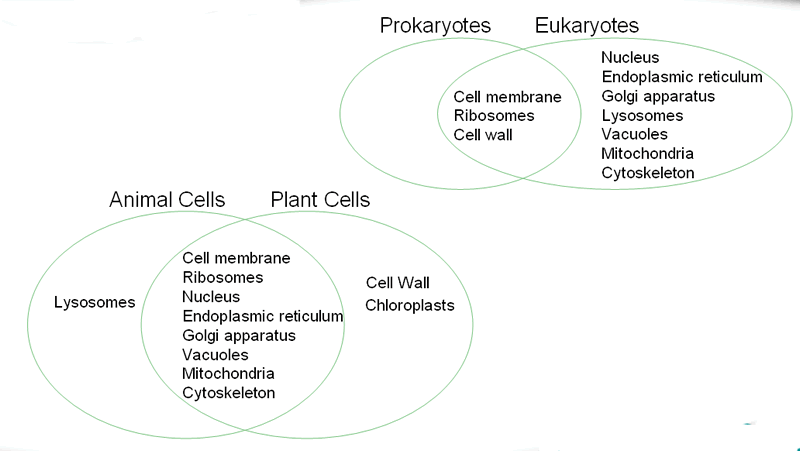
Electrons are distributed throughout the atom, while in later models they are in orbitals. Thomsons atom contains no nucleus and instead has a sea of positive charge, rather than a central positive charge.